
HPS Pharmacies wish to give notice that Generic Health, in consultation with the Therapeutic Goods Administration (TGA), has issued a product alert Fentanyl GH Solution for Injection as follows:
Fentanyl GH Solution for Injection
Fentanyl 500 microgram/10 mL
ARTG 201871
This product alert was initiated following a report of one ampoule that was found to be damaged within a sealed blister. There was a hole in the tip of the ampoule and a small amount of liquid in the blister casing. Generic Health advises that the defect can be clearly identified. However, a residual risk of fragile ampoule heads with poorly visible damage cannot be fully excluded.
No patient harm has been reported due to this issue.
The only batch affected by this notice is: 40679 (Exp October 2027)
Please inspect each ampoule prior to use. Any ampoules that show signs of damage or structural irregularity should not be used.
Retain this notice in a prominent position, including in other related business units for one month. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact Generic Health on 03 9780 9700 or your pharmacist at HPS Pharmacies.
Download PDF

HPS Pharmacies wish to advise of a supply interruption for codeine linctus as follows:
Actacode Oral Liquid
Codeine phosphate hemihydrate 5mg/mL
ARTG 124245
Gold Cross Codeine Linctus
Codeine phosphate hemihydrate 5mg/mL
ARTG 27219
Normal supplies are expected to resume by the end of March 2026.
Retain this notice in a prominent position, including in other related business units until supplies return to normal. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact your pharmacist at HPS Pharmacies.
Download PDF

HPS Pharmacies wish to give notice that Bayer Australia, in consultation with the Therapeutic Goods Administration (TGA), has issued a product recall for Gastrografin® Oral Liquid and Urografin® Injection as follows:
| Product |
ARTG |
Affected batches |
| Gastrografin® (with star anise oil) |
10684 |
MA04JPZ (Exp 30-Apr-2026)
MA04NJF (Exp 30-Nov-2026) |
| Gastrografin® (without star anise oil) |
471827 |
MA04T47 (Exp 30-Apr-2029)
MA04T49 (Exp 30-Apr-2029) |
| Urografin® 30% 10mL ampoules |
42161 |
MA04F5E (Exp 30-Jun-2026)
MA040FA (Exp 30-Jun-2026)
MA047MV (Exp 30-Jun-2026)
MA04KBH (Exp 30-Jun-2026)
MA04BVS (Exp 30-Nov-2026)
MA04S7C (Exp 28-Feb-2030)
MA04V62 (Exp 28-Feb-2030)
MA04VUM (Exp 28-Feb-2030) |
| Urografin® 30% 250mL bottle |
42163 |
MA03X2V (Exp 31-Jul-2026)
MA04MKJ (Exp 28-Feb-2029)
MA04MKK (Exp 31-Jul-2029)
MA04RNS (Exp 31-Jul-2029)
MA04SJ2 (Exp 31-Jul-2029) |
This product recall was initiated following identification of the nitrosamine impurity, N‑Nitroso‑Meglumine (NO-meglumine), at levels exceeding the calculated applicable intake limits.
Please inspect your stock and quarantine all products from the affected batches. If HPS Pharmacies provide imprest management services to your facility, an HPS representative will inspect all stock stored in the designated imprest areas at the earliest opportunity. All affected products will be quarantined for return to the supplier. Alternative stock is not currently available. An HPS Pharmacies staff member will contact senior medical and nursing staff if there is an immediate impact to your facility.
Retain this notice in a prominent position, including in other related business units for one month. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact Bayer Australia 1800 633 723, or your pharmacist at HPS Pharmacies.
Download PDF

HPS Pharmacies wish to advise that all brands of betahistine 16mg tablets are experiencing a supply interruption as follows:
| Product |
ARTG |
Estimated return date |
| Betahistine Lupin |
388870 |
Early March 2026 |
| Betahistine Sandoz |
253732 |
Late April 2026 |
| Betahistine Viatris |
286806 |
End-March 2026 |
| Serc® |
61687 |
End-March 2026 |
Setear® (betahistine 24mg) tablets are currently available in most warehouses, although some states are also experiencing temporary supply issues.
Retain this notice in a prominent position, including in other related business units until supplies return to normal. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact your pharmacist at HPS Pharmacies.
Download PDF

HPS Pharmacies wish to give notice that Sandoz, in consultation with the Therapeutic Goods Administration (TGA), has issued a product alert for Oxycodone Sandoz Modified Release Tablets as follows:
Oxycodone Sandoz Modified Release Tablets
Oxycodone hydrochloride 5 mg
ARTG 153605
This product alert was initiated following reports that a batch of modified release tablets was supplied without the ‘modified release’ text on the blister pack. This may cause confusion between other forms of oxycodone tablets and lead to medication errors. The outer carton correctly describes the dosage form as modified release.
The only batch affected by this notice is: PS9028 (Expiry: Feb 2028)
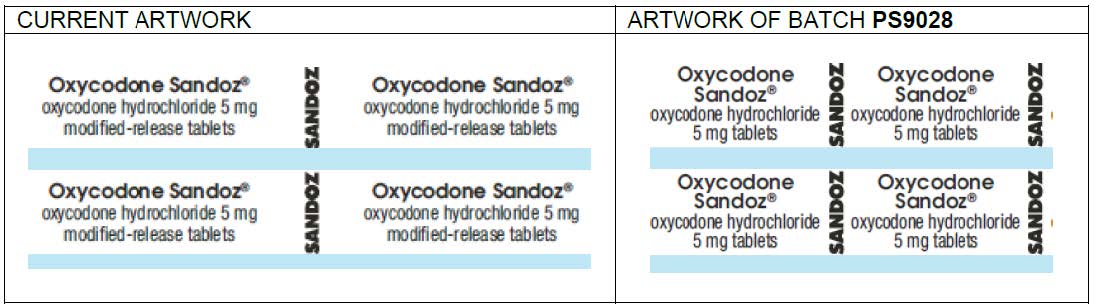
Actions recommended by Sandoz:
- Reassure patients that the product is modified release as described in the outer packaging.
- Store medications in their original packaging.
Sandoz advises that product with the corrected blister packaging will be available from September 2026.
Retain this notice in a prominent position, including in other related business units for one month. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact Sandoz at 1800 726 369 or your pharmacist at HPS Pharmacies.
Download PDF

HPS Pharmacies wish to give notice that Sanofi-Aventis is discontinuing Epilim® Crushable tablets as follows:
Epilim® Crushable Tablets
Sodium valproate 100mg
ARTG 15373
Epilim® Crushable tablets are set to be deleted from the Australian market on 28th February 2026. Alternative presentations of sodium valproate will remain available.
Retain this notice in a prominent position, including in other related business units for one month. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact Sanofi-Aventis on 1800 818 806 or your pharmacist at HPS Pharmacies.
Download PDF

HPS Pharmacies wish to advise that Pfizer is experiencing a supply interruption for Dantrium® vials as follows:
Dantrium® Vials
Dantrolene sodium hemiheptahydrate 20mg
ARTG 14435
Normal supplies are expected to resume by the end of February 2026.
An internationally registered alternative of dantrolene 20mg vials has been approved for supply under Section 19A of the Therapeutic Goods Act 1989. Stock may also be accessed via the Special Access Scheme (SAS), if required. The estimated lead time for these products is 15 days.
Retain this notice in a prominent position, including in other related business units until supplies return to normal. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact Pfizer on 1800 675 229, or your pharmacist at HPS Pharmacies.
Download PDF

Further to DrugAlert vol 1041, HPS Pharmacies wish to provide an update on the availability of Palexia® IR as follows:
Palexia® IR 50mg Tablets
Tapentadol (as hydrochloride) 50 mg
ARTG 165310
The short-term supply issue related to delivery delays has now been resolved. Wholesalers have received stock and are currently processing backorders. Normal supplies are expected to be received from wholesalers starting from 23rd January 2026.
Retain this notice in a prominent position, including in other related business units until supplies return to normal. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact Seqirus on 1800 008 275, or your pharmacist at HPS Pharmacies.
Download PDF

HPS Pharmacies wish to advise that Seqirus is experiencing a supply interruption for Palexia® IR as follows:
Palexia® IR 50mg Tablets
Tapentadol (as hydrochloride) 50 mg
ARTG 165310
This supply interruption is due to a delay in delivery from the manufacturer. Normal supplies are expected to resume by late January to early February 2026. Consult prescribers for suitable alternative opioids during this shortage.
Retain this notice in a prominent position, including in other related business units until supplies return to normal. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact Seqirus on 1800 008 275, or your pharmacist at HPS Pharmacies.
Download PDF

HPS Pharmacies wish to advise that Pfizer is experiencing a supply interruption for Prostin® E2 as follows:
Prostin® E2 Vaginal Gel
Dinoprostone 1mg
ARTG 9983
Prostin® E2 Vaginal Gel
Dinoprostone 2mg
ARTG 9984
Normal supplies are expected to resume by the end of June 2026.
An internationally registered alternative of each strength has been approved for supply under Section 19A of the Therapeutic Goods Act 1989. However, these products are not yet available for immediate supply. Updates will be provided when more information becomes available.
No equivalent products are currently available. Potential alternatives that prescribers may consider include dinoprostone controlled-release pessary (Cervidil®), oxytocin, and misoprostol. Availability of these products may vary throughout the country.
Retain this notice in a prominent position, including in other related business units until supplies return to normal. Report any problems identified with medicines, vaccines or medical devices to the TGA.
Should you require further information regarding this matter, please contact Pfizer on 1800 675 229, or your pharmacist at HPS Pharmacies.
Download PDF




